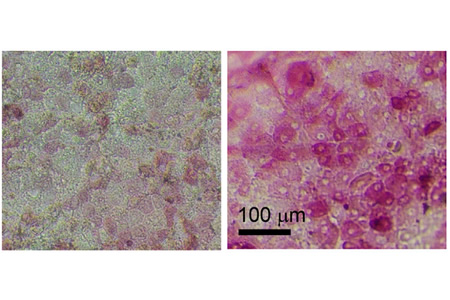
Scientists in Japan have successfully differentiated enterocytes from human induced pluripotent stem cells (HiPSCs) which can be used to create a more accurate model of the human small intestine.
An accurate, in vitro model of the small intestine is important for studying drug transport and metabolism. Many drugs are administered orally and are absorbed into the blood stream via the small intestine. To determine how well novel drugs are absorbed, an in vitro or animal model is required.
One of the most commonly used cell lines in research to model the small intestine is the human colon cancer cell line Caco-2. Caco-2 cells are used to model the small intestine endothelium, but there are flaws with this model as the cells are derived from cells of the colon. Consequently, they differ from the cells of the small intestine in important ways. One important difference is Caco-2 cells do not express cytochrome P450 3A4 (CYP3A4), which is found in abundance in the cells of the small intestine. The CYP3A4 protein plays a critical role in drug metabolism. In addition, Caco-2 cells have high cell-line to cell-line variations.
To create a more accurate model of the small intestine, researchers at Tokyo Institute of Technology, University of Tokyo, Kanto Chemical Co. Inc., Shionogi & Co., Ltd. and Shionogi TechnoAdvance Research Co., Ltd., used a procedure developed using embryonic stem cells and applied this to hIPSCs. Using this new technique, the researchers generated intestinal progenitor cells, which resembled the early stages of the cells of the small intestine. The intestinal progenitor cells were cultured on a collagen vitrigel membrane (CVM) and were treated with 6-bromoindirubin-3′-oxime, dimethyl sulfoxide, dexamethasone, and activated vitamin D3, to mature the cells.
The mature cells more closely resembled the enterocytes found in the small intestine and, in contrast to Caco-2 cells, they expressed CYP3A4 and efflux transporter proteins that regulate drug absorption.
“The hiPSC-derived enterocyte-like cells established in this study could be used for the quantitative prediction of the intestinal absorption of drugs in humans under special occasions such as alteration of the functions of transporters/metabolic enzymes by drug-drug interactions as well as normal conditions,” said Dr. Shoen Kume, Professor at Tokyo Institute of Technology, and co-corresponding author of the study.
You can read more about the study in the paper – Generation of human-induced pluripotent stem cell-derived functional enterocyte-like cells for pharmacokinetic studies– which was recently published in the Stem Cell Reports. DOI: 10.1016/j.stemcr.2020.12.017